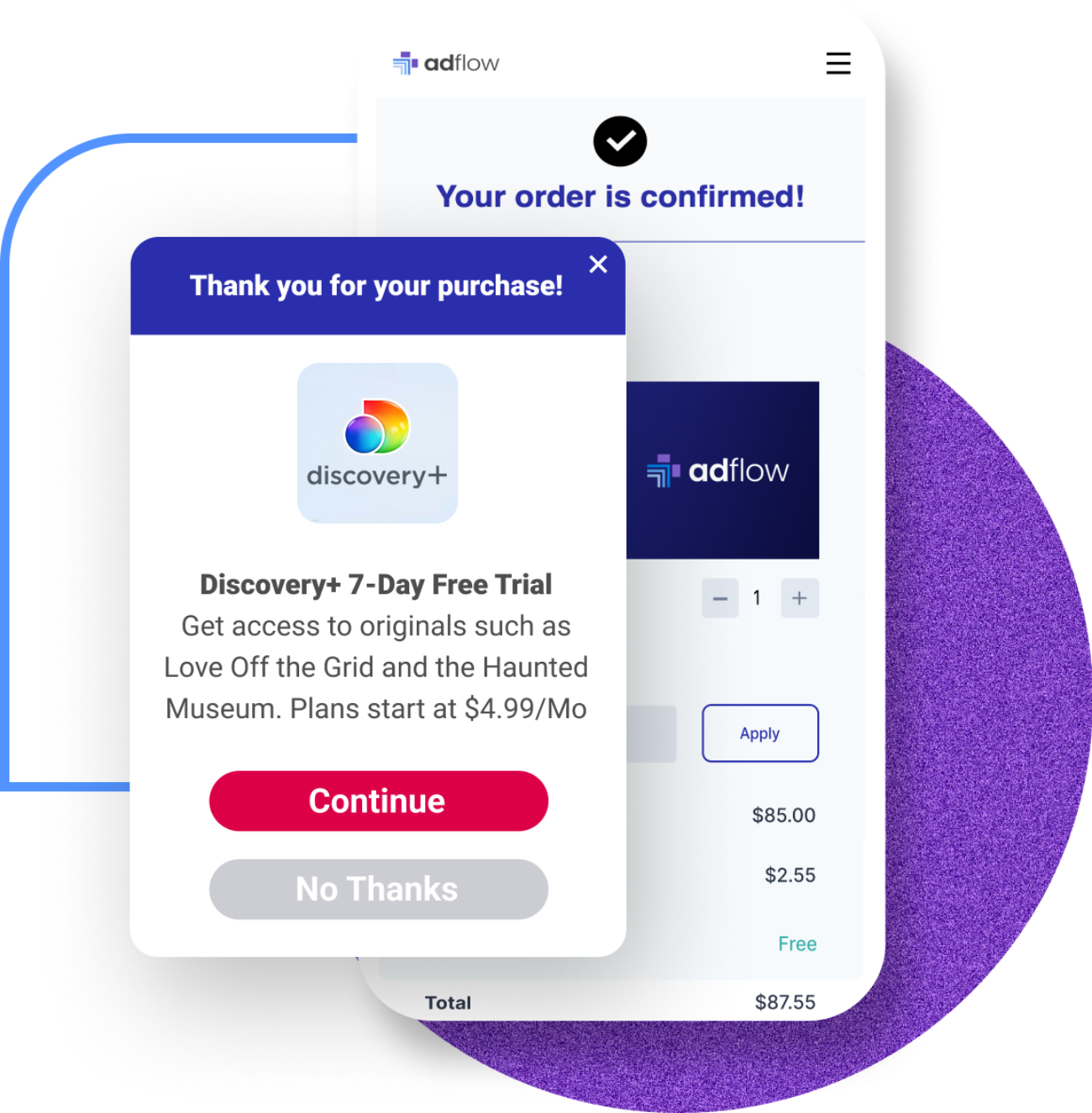
Upgrade Your
Acquisition
Growth
Audience
Strategy
Leading Brands Trust Fluent to Power their Marketing Strategy















Solutions
Advertisers partner with Fluent to explore and optimize alternative user-acquisition channels
Publishers team up with Fluent to maximize their ad inventory and drive more revenue from their unique audiences
We’ll Help You Connect and Convert Audiences Primed for Acquisition
01 Target
Leverage Fluent’s robust modeled data to enhance your targeting and deliver relevant offers.
02 Connect
Reach hundreds of millions of monthly active users, ready to discover new products and services.
03 Acquire
Convert pre-qualified audiences with an active interest in your brand.
04 Retain
Drive continued engagement and boost LTV with down-funnel conversion events.
05 Optimize
Partner with our team of experts to share conversion feedback and optimize ROAS.